Researchers created a 3D environment for stem cells using a stiffness-gradient hydrogel. AFM modulus measurements helped determine correlations between cell and nuclear volume and mechanosensitive response.
Mechanotransduction, in which cells respond electrochemically to mechanical cues, is essential to processes ranging from disease to the sense of hearing. Although most cell biology experiments use a 2D geometry, mechanotransduction behavior in 3D—the true state of cells in vivo—can differ from that in 2D.
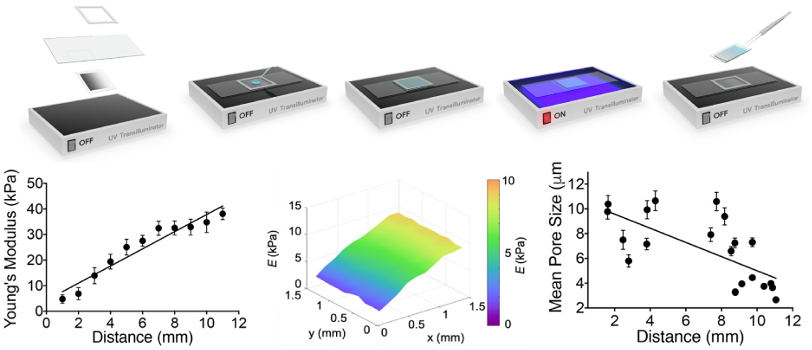
To explore this issue, a team led by researchers at the University of Western Australia used a gelatin methacryloyl (GelMA) hydrogel whose stiffness could be altered by UV polymerization. AFM modulus measurements verified the samples had a linear stiffness gradient over physiologically relevant values (5-38 kPa).
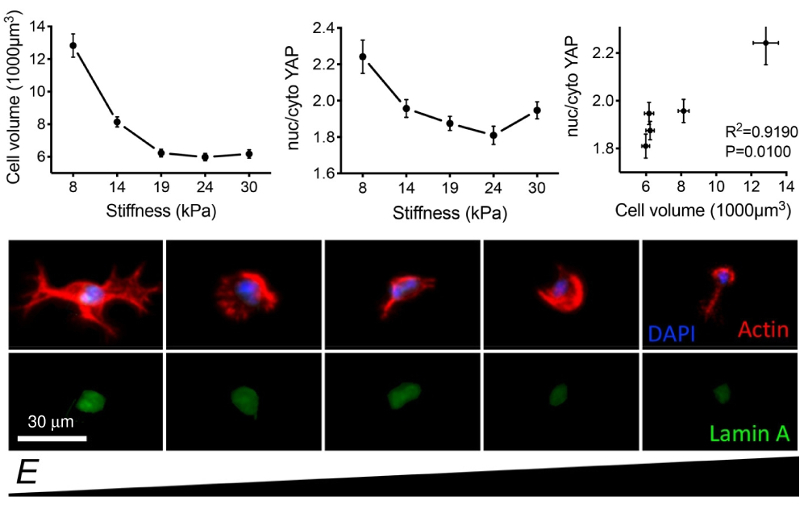
Researchers encapsulated adipose-derived stem cells in the hydrogel and characterized their morphology and response to mechanosensitive proteins. Stiffer regions showed smaller nuclear and cellular volumes, and reduced mechanotransduction including protein expression and localization. In more compliant regions, the trends were reversed.
Together with other findings, these results suggest that volume adaptation to stiffness plays an important role in 3D mechanotransduction. They also highlight the need for further 3D studies to better understand mechanosensitive response in vivo.
Instrument used
MFP-3D
Techniques used
Hydrogel stiffness was measured by force curve techniques on an MFP-3D AFM. Measurements were performed with the hydrogels immersed in phosphate buffer saline (PBS) solution using cantilevers containing gold-coated, pyramid-shaped tips. MFP-3Ds offer the highest performance and lowest-noise force measurements at a low price point. Data fitting to determine Young’s modulus was simplified through Asylum’s built-in analysis software. For bioscience experiments such as these that combine AFM and optical microscopy, the MFP-3D BIO offers an integrated platform with uncompromised performance.
Citation: L. Major, A. Holle, J. Young et al., Volume adaptation controls stem cell mechanotransduction. ACS Appl. Mater. Interfaces 11, 45520 (2019). https://doi.org/10.1021/acsami.9b19770
Note: The data shown here are reused under fair use from the original article, which can be accessed through the article link above.



