Tags: AFM, Atomic Force Microscopy, contact mode, force curves, Scanning Probe Microscopy, SPM, tapping mode
Electrochemical AFM experiments and molecular dynamics simulations allowed researchers to characterize the nanoscale structure of electrical double layers formed by ionic liquids at a solid interface.
 Batteries, supercapacitors, and other energy storage technology could benefit greatly from the unique properties of ionic liquids. However, commercial implementation is hindered by limited knowledge about the electrical double layer (EDL) formed at interfaces between ionic liquids and solids.
Batteries, supercapacitors, and other energy storage technology could benefit greatly from the unique properties of ionic liquids. However, commercial implementation is hindered by limited knowledge about the electrical double layer (EDL) formed at interfaces between ionic liquids and solids.
To address this issue, researchers in the U.S. and China investigated EDL structure and dynamics using in-situ electrochemical AFM experiments and molecular dynamics simulations. They examined EDLs formed by 1-butyl-1-methylpyrrolidinium bis-(trifluoromethanesulfonyl)imide (PYR14-TFSI) on highly ordered pyrolytic graphite (HOPG).
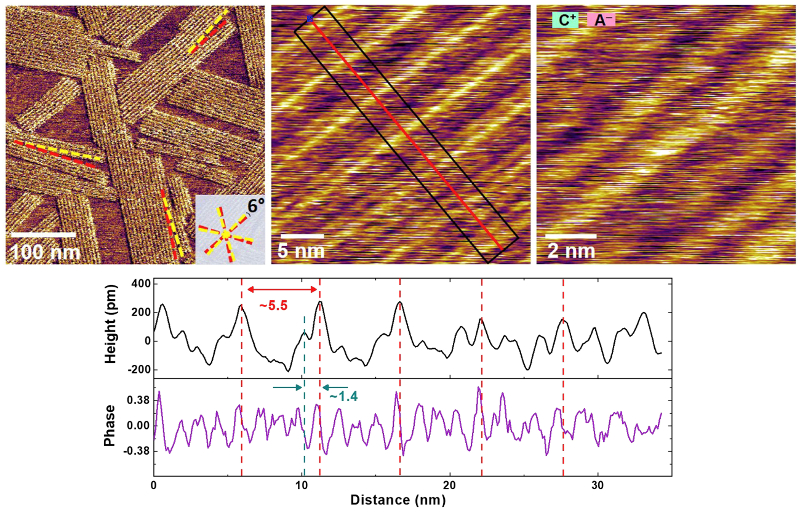
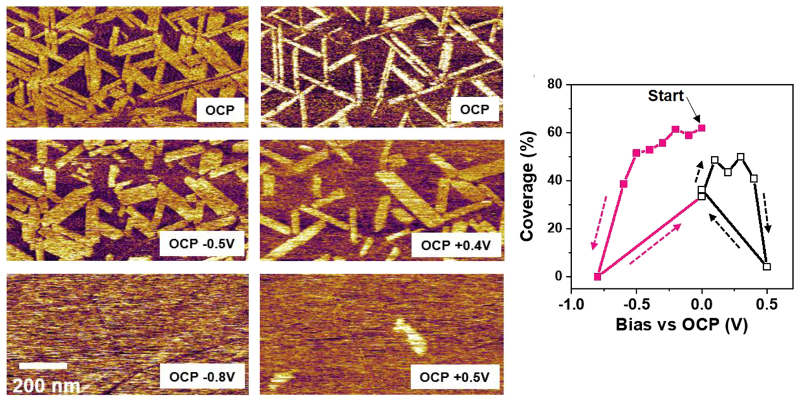
High-resolution AFM images of the EDL in-plane structure revealed the first adsorbed ion layer contained both disordered and ordered lateral domains with nanoscale periodicity. The coverage of the lateral domains was found to decrease and then vanish when a positive or negative electrical bias relative to open circuit potential (OCP) was applied. These findings were interpreted with molecular dynamics simulations that considered a bimodal distribution of anion orientations.
The results increase our understanding of EDLs and may provide the insight into charge and discharge kinetics of ionic liquids needed for use in electrochemical applications.
Instrument used
Cypher
Techniques used
The EDL structure was imaged using a Cypher AFM with the cantilever immersed in droplet of PYR14-TFSI. Images in the top figure represent tapping mode phase (left) and height (middle and right) and demonstrate the superb resolution possible with Cypher AFMs, even when operating in liquid. Prior to imaging, force measurements were made to determine the ionic liquid out-of-plane layering positions and appropriate imaging parameters. The bottom figure shows deflection images acquired while an electrical bias was applied to the HOPG substrate. Although not used in this work, the Electrochemical Cell accessory is a convenient turnkey solution for in-situ electrochemical studies.
Citation: W. Tsai, J. Come, W. Zhao et al., Hysteretic order-disorder transitions of ionic liquid double layer structure on graphite. Nano Energy 60, 886 (2019). https://doi.org/10.1016/j.nanoen.2019.04.022
Note: The data shown here are reused under fair use from the original article, which can be accessed through the article link above.


 Batteries, supercapacitors, and other
Batteries, supercapacitors, and other 